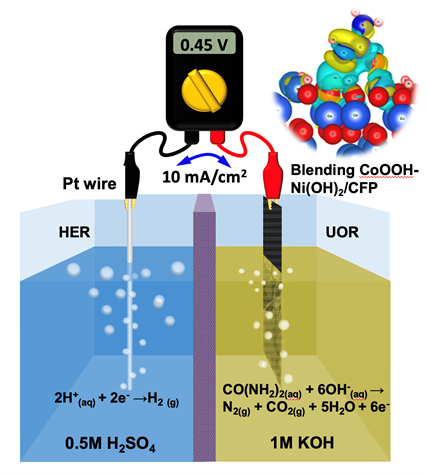
09 Aug. 2024, our work “Boosted Urea Electrooxidation Activity by Dynamic Steady Blending CoOOH-Ni(OH)2 Nanoclusters for H2 Production in a pH-asymmetric Electrolyzer” has been accepted by J. Mater. Chem. A
Boosted Urea Electrooxidation Activity by Dynamic Steady Blending CoOOH-Ni(OH)2 Nanoclusters for H2 Production in a pH-asymmetric Electrolyzer
Shih-Mao Peng,1,2# Shu-Ting Chang,2# Chia-Che Chang,3# Priyadarshini HN,2 Chun-Chih Chang,4* Kuan-Chang Wu,2 Yung-Hung Huang,5 Yi-Chia Chen,5 Tsung-Rong Kuo,6 Chih-Wen Pao,3 Jeng-Lung Chen,3 Di-Yan Wang5*
https://doi.org/10.1039/D4TA04241G
Electrochemical urea oxidation reaction (UOR) is a promising alternative to the oxygen evolution reaction for reducing the overall potential of the hydrogen evolution reaction during water electrolysis. The theoretical potential for the UOR is only 0.37 V versus reversible hydrogen electrode (RHE). However, the kinetics of the six-electron transfer process involved in the UOR are inherently sluggish, resulting in high overpotential during the reaction. This study designed an active catalyst with a lower kinetic barrier in the UOR by fabricating blending CoOOH–Ni(OH)2 nanoclusters through the structural transformation of amorphous Co–Ni hydroxide films. This structural transformation was investigated using high-angle annular dark-field scanning transmission electron microscopy, corresponding energy-dispersive X-ray spectroscopy, and in situ X-ray absorption spectra. The blending CoOOH–Ni(OH)2 nanoclusters exhibited superior electrocatalytic activity in the UOR in an alkaline environment, achieving a low onset potential of 1.24 V (vs. RHE) in 1 M KOH with 0.5 M urea. We employed the CoOOH–Ni(OH)2 nanoclusters as anodic electrocatalysts in a two-cell electrolyzer for asymmetric electrocatalysis. Hydrogen could be produced at a remarkable current density of 10 mA/cm2 at a low applied potential of only 0.45 V. Density functional theory calculations revealed that blending CoOOH–Ni(OH)2 nanoclusters with more oxygen vacancies exhibited a lower Gibbs free energy for the intermediate reaction pathway of NCONH2 → NCONH, compared with the fine structure of CoNiOx (x = 2–3). This study lays down a novel pathway for developing new blending electrocatalysts to be used in electrochemical reactions.
2024
| 88 | Yi-Chia Chen, Kuan-Chang Wu, Jou-Chun Lin, Anupriya Singh, Yu-Dian Chen, Hsin-An Chen, Di-Yan Wang*, 2024: Discovery of Thermodynamic-Control Two-Dimensional Cs6Pb5I16 Perovskite with Unique Green Emission Color via Dynamic Structural Transformation, J. Phys. Chem. Lett., 2024, 15, 9311−9318. |
| 87 | Shih-Mao Peng,Shu-Ting Chang, Chia-Che Chang, H.N. Priyadarshini, Chun-Chih Chang,* Kuan-Chang Wu, Yung-Hung Huang, Yi-Chia Chen, Tsung-Rong Kuo,Chih-Wen Pao, Jeng-Lung Chen, Di-Yan Wang*, 2024: Boosted Urea Electrooxidation Activity by Dynamic Steady Blending CoOOH-Ni(OH)2 Nanoclusters for H2 Production in a pH-asymmetric Electrolyzer, J. Mater. Chem. A, 2024, 12, 24126–24135. |
| 86 | Chia-Che Chang, Yi-Chia Chen, Kuan-Chang Wu, H.N. Priyadarshini, Lo-Yu Lee, Jeng-Lung Chen, Chang-Ru Lee, Chih-Wen Pao* and Di-Yan Wang*, 2024: Subnanometer-sized CuOx Clusters on TiO2 as Active Photocatalysts for Ammonia Production from Photocatalytic Nitration Reduction Reaction, ChemCatChem, 2024, 16, e202400596. |
Priyadarshini HN
Priyadarshini HN
ngowdapriya@gmail.com
National Taiwan Normal University
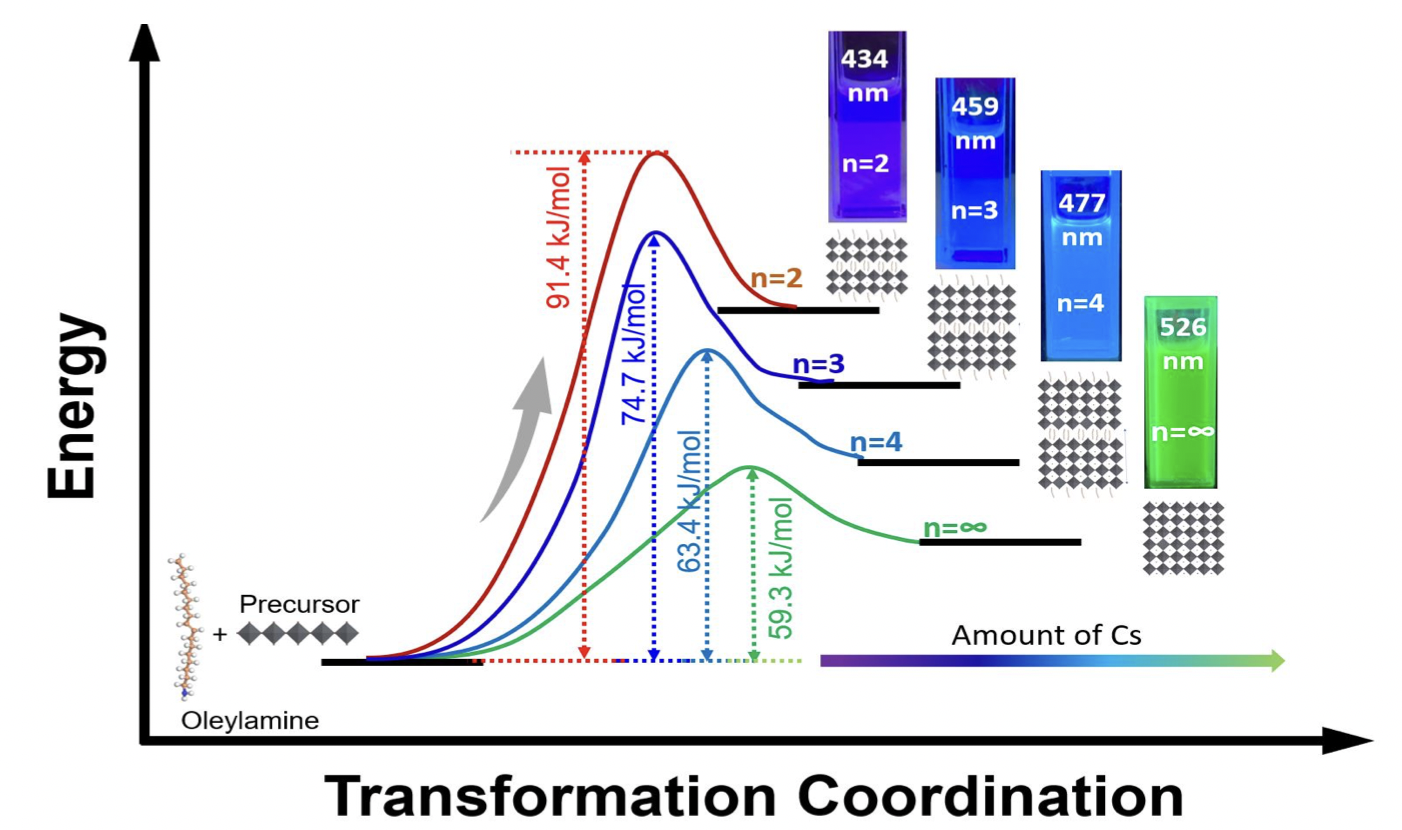
11 Jul. 2023, our work “Kinetic Studies of Oleylamine based 2D-Lead Bromide Perovskite with Controllable n-value by Sequential Addition of Cesium at Room Temperature” has been accepted by Journal of Physical Chemistry C
Invited Article in The Physical Chemistry of Perovskites VSI Virtual Special Issue.
Kinetic Studies of Oleylamine based 2D-Lead Bromide Perovskite with Controllable n-value by Sequential Addition of Cesium at Room Temperature
Anupriya Singh, Yi-Chia Chen, Kuan-Chang Wu, Shih-Mao Peng, Tsung-Rong Kuo, and Di-Yan Wang*
https://doi.org/10.1021/acs.jpcc.3c04038
The outstanding properties of lead-based perovskite nanomaterials have led researchers to investigate the potential of their two-dimensional (2D) perovskite counterparts with the chemical formula A’2An+1BnX3n+1. Despite their advantages such as better stability and higher exciton binding energy, the synthesis of phase pure 2D perovskite nanomaterials remains challenge due to the fast nucleation process. Here, we demonstrate a simple and efficient method to synthesize 2D perovskite nanomaterials with phase pure specific n-values. The room temperature nucleation and growth of perovskites with specific n-values are controlled in a two-step synthesis method by the optimized amount of cesium. Our 2D perovskite nanomaterials with specific n-values exhibit high-purity photoluminescence (PL) peaks in the range from deep-blue to azure-blue emission color. X-ray diffraction (XRD) and attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR) were used to elucidate the kinetic reaction of oleylamine with PbBr2 and the effect of cesium addition in formation of 2D perovskite nanomaterials. In-situ photoluminescence studies were also performed to calculate the activation energy (Ea) of different specific n-value nanomaterials by using the Arrhenius equation. This study not only helps to visualize the effect of addition of Cs amount on the stacking of monolayers of [PbBr6]4-octahedrons, but also provides an easier way to control the n-value in 2D perovskite nanomaterials.