Hydration Shell Modulation of Al3+ for Efficient Aluminum Electrodeposition and Suppressed Hydrogen Evolution in Aqueous Batteries
Jhen-Hao Huang,# Meng-Chi Hsieh,# Swathi M. Gowdru,# Chia-Che Chang,# Chen Chen,# Shu-Yao Huang, Yung-Yi Huang, Zhi-Jie Li, Yu-Cheng Wu, Han-Hsuan Hsieh, Yi-Chia Chen, Chun-Chih Chang*, Ying-Huang Lai, Chun-Jern Pan, and Di-Yan Wang*
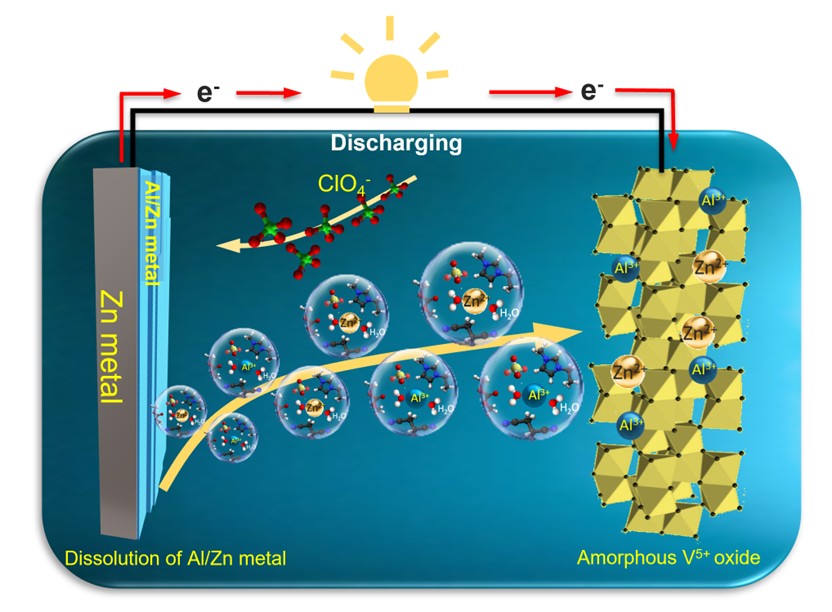
Reducing free water in aqueous electrolytes is crucial for suppressing hydrogen evolution reaction (HER) and improving the electrochemical performance of aluminum plating. In this study, an electrolyte system (Al(ClO4)3-DWES) was designed by introducing organic molecules dimethyl carbonate, 1-ethyl-3-methylimidazolium cation, and succinonitrile to modulate hydration shell and eliminate free water. Raman spectroscopy and molecular dynamics (MD) simulations confirmed the HER suppression through enhanced hydration bonding during Al plating. The galvanostatic cycling scan demonstrated the superior performance of Al(ClO4)3-DWES in enabling uniform Al deposition and improving plating/stripping efficiency on zinc foil. The detailed characterizations validated that the smooth and dense Al metal was actually plated with Zn metal, because Zn ions was found to be dissolved into electrolyte at the beginning cycles. Moreover, Al(ClO4)3-DWES electrolyte was successfully employed in aqueous Al-Zn/amorphous vanadium(V) oxide batteries, exhibiting high discharging capacity (250 mAh/g), excellent rate capability and superior cycling stability over 750 cycles with 99% Columbic efficiency. X-ray photoemission spectra revealed the roles of Al3+ and Zn2+ ions in the reversible intercalation/deintercalation process in the cathodic materials. Overall results confirm that hydration shell modulation of Al³⁺ with eliminating free water in the electrolyte enhances Al plating and enables stable Al-ion battery operation, paving the way for advanced aqueous battery technologies.